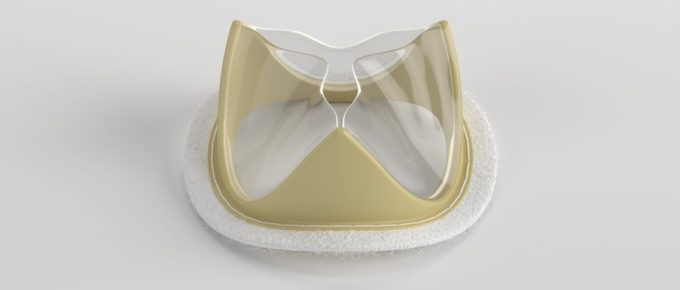
SALT LAKE CITY, Utah – August 1, 2023 - Foldax®, Inc., a leader in the development of innovative, polymer heart valves, today announced that the first 30 patients have been treated outside of the U.S. …
News
Enable Injections Receives First U.S. Food and Drug Administration (FDA) Approval
CINCINNATI, Oct. 2, 2023 /PRNewswire/ -- Enable Injections, Inc. ("Enable") today announced that the U.S. Food and Drug Administration (FDA) has approved the EMPAVELI Injector (enFuse®) for the …
Brixton Biosciences’ Coolio™ Therapy Granted Breakthrough Device Designation by the FDA
CAMBRIDGE, Mass., Nov. 13, 2023 /PRNewswire/ -- Brixton Biosciences, a clinical-stage life sciences company developing novel therapies for chronic and acute pain, is delighted to …
MuReva Phototherapy presents data from pilot study of Head and Neck Cancer Patients Treated Daily with an Intraoral Photobiomodulation (PBM) Device during the ASTRO 2023 Annual Meeting
MuReva Phototherapy presented data from a pilot prospective study of the company’s Intraoral Photobiomodulation (PBM) Device during the the American Society for Radiation Oncology’s (ASTRO) 65th …
S4 Medical Raises Financing for Esolution Esophageal Deflection Device in AF Ablation Procedures
August 4, 2023—S4 Medical Corp, which is developing an esophageal deflection device to use during atrial fibrillation (AF) ablation procedures, recently announced the closing of a $5 million …
S4 Medical Gets FDA Authorisation for Esophageal Deviation Instrument
S4 Medical Corporation is dedicated to enhancing safety in atrial ablation treatments and stated that the U.S. FDA had granted de novo certification for their esolution® esophageal deviation …
Continue Reading about S4 Medical Gets FDA Authorisation for Esophageal Deviation Instrument →